“Fluid Bed Dryer (FBD) in Pharma: Working Principle + 7 Common Problems & Solutions”
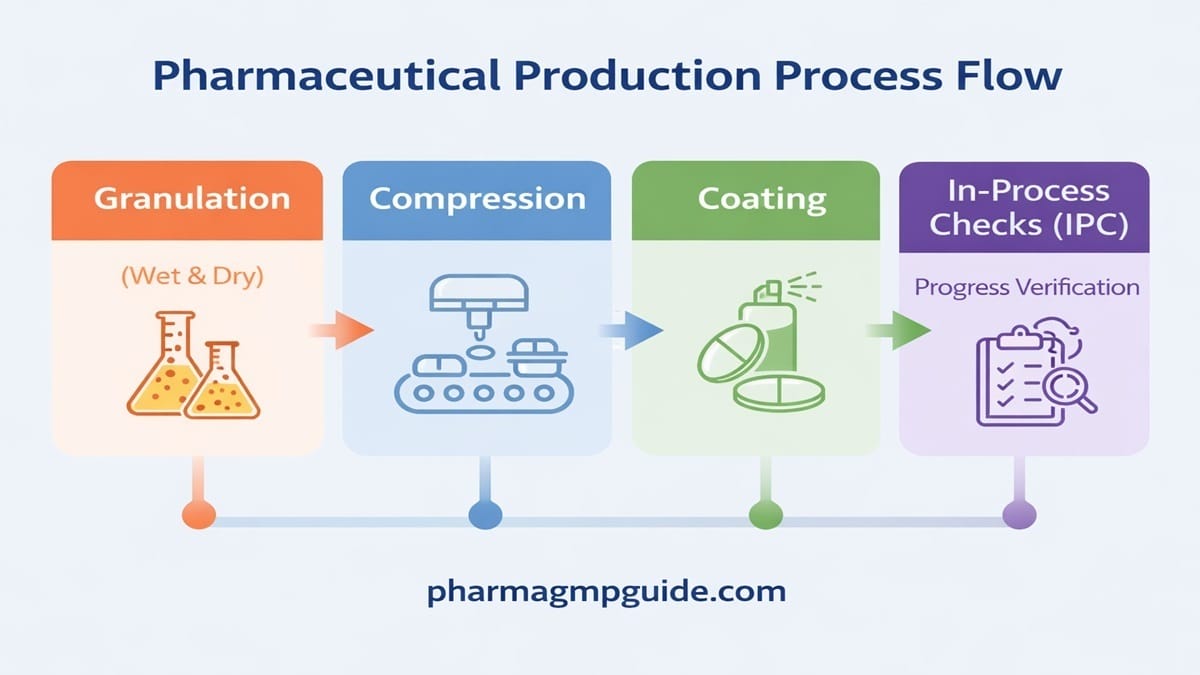
Introduction If you’ve ever worked in a granulation area, you already know one thing—drying is where most mistakes happen. Wet granules look perfect, but if drying goes wrong: That’s where the Fluid Bed Dryer (FBD) becomes a game-changer. Unlike tray drying (slow and uneven), FBD gives:✔ Fast drying✔ Uniform moisture removal✔ Better product quality In […]