Capsule Manufacturing Process: 9 Powerful Tips to Avoid Critical GMP Mistakes
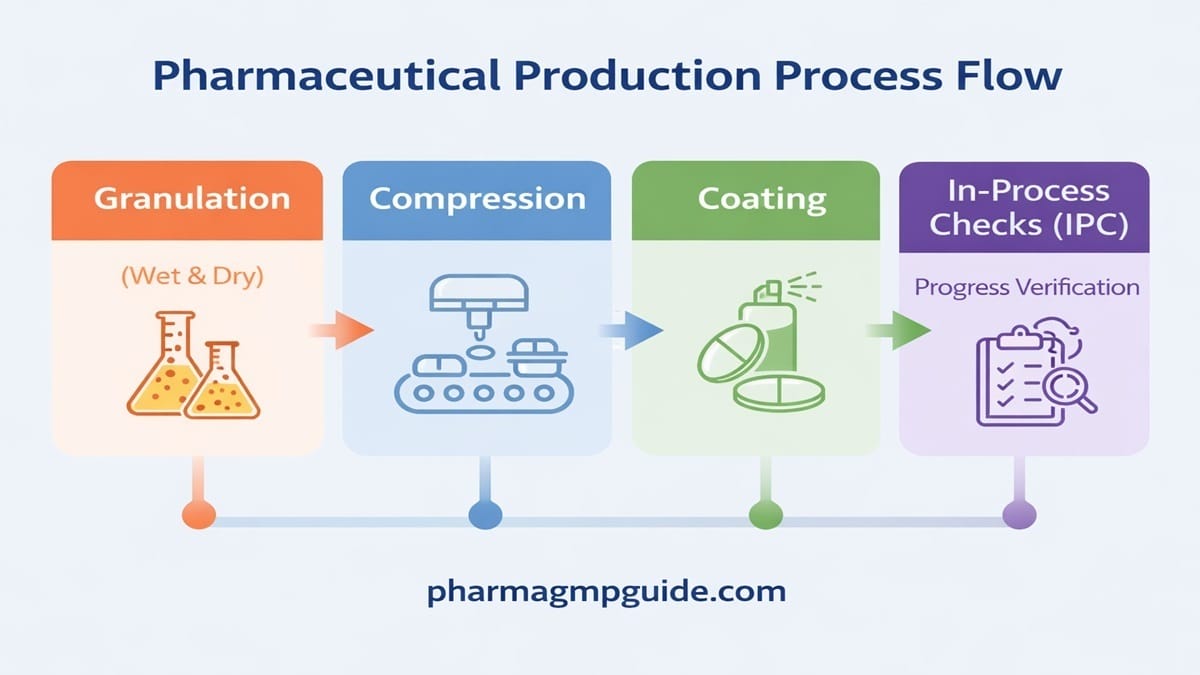
Introduction Capsules are one of the most commonly used oral dosage forms in the pharmaceutical industry because they are easy to swallow, provide accurate dosing, and improve patient compliance. The capsule manufacturing process involves multiple controlled steps such as material preparation, filling, sealing, inspection, and packaging. In real pharmaceutical manufacturing, capsule production is not just […]